iT-BaSTAH – Training-app to perform programmed sports therapy for people with haemophilia (April 2018 – October 2018)
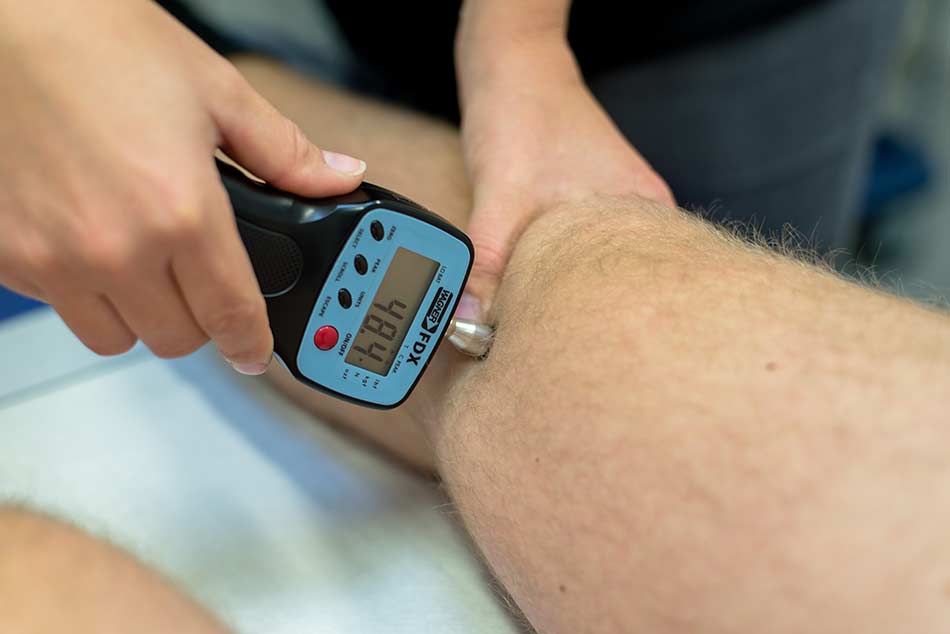
Several studies demonstrate the positive influence of specific training on performance and quality of life of people with haemophilia. This is why nowadays specific movement therapeutic measures form an inherent part of treatment principles for people with haemophilia. Many patients, though, do not have the possibilities to participate in these kinds of therapies. In our current project we would like to modernise PST by making it accessible to as many people with haemophilia as possible. During home training, therapists and patients permanently communicate in order to motivate, respond to questions and continuously adapt the training plan to the patients’ necessities. Up to now, home training has been guided by an exercise catalogue and training plans in paper form or computer-based. While the exercise catalogue can be accessed via our website, the exchange of training plans takes place via e-mail or mailing route. Likewise, communication between patient and therapist happens via e-mail and telephone. Due to the high and time-consuming supervision expenditure, only a limited number of patients have been able to be guided. Additionally, using paper forms in the digital age simply seems to not be contemporary anymore. This is why a sport-therapeutic training-app for people with haemophilia, whose applicability and effectiveness will be tested in a 12-week study, is supposed to be developed by means of our current project. The app is developed to be as patient-oriented as possible through a demand analysis to optimally fulfill the target group’s needs and standard. The demand analysis was conducted in the sports camp in April 2018. Once the training-app is developed, it shall enable every user to access the app via a password-protected account on a computer, tablet or smartphone.
The training-app contains an individualized training plan as well as descriptions and instructions for exercise performance. Furthermore, general training plans and exercises classified according to their intensity and demand are made available, so that the user can individually design and vary training. Every user gets the possibility to use a reminder function as well as to record every training session to provide the therapist with information in order to get feedback. This direct feedback enables individual to respond to individual matters more quickly, such as answering questions or setting changes in the training plan. In order to test the applicability and at the same time, effectiveness of the training-app, a 12-week pilot study is supposed to evaluate these aspects. After a control phase of six weeks without using the app, the participants perform a six-week training intervention guided by the app. At three points in time in total (1. at the beginning of control phase, 2. at the end of control phase = beginning of intervention phase, 3. at the end of intervention phase), we would like to record subjective physical capacity, quality of life, physical activity and the situation of pain by means of standardized questionnaires. Furthermore, participants are asked to document their physical activity as well as bleeding episodes and factor substitution throughout the course of the study. In order to create a training plan that is individually adjusted to the patient’s physical situation and needs, it is essential to capture the general physical situation as well as the joint status in a personal conversation and a check via joint score. At the same time, participants receive a detailed introduction to the usage of the training-app and theoretical and practical instructions in order to perform training. Investigation and instruction will take place in the sports camp in October 2018.
The project iT-BaSTAH is conducted in cooperation with the Department of Sports Medicine and Haemostaseological Ambulance of the Medical School of Hannover and supported by the company Pfizer.
A successful implementation of this kind of app could enable a great number of people with haemophilia to perform a guided and individualized home training in order to substantially improve their joint status and personal well-being as well as to increase their general quality of life.
If you are interested in participating in this pilot study to evaluate the training-app we would like to invite to contact us:
Study coordinator: Marie Herzig
Phone +49 (0)202 - 439 5915
E-mail herzig@uni-wuppertal.de
Haem-Pain-Profile (March 2017 - March 2019)
The knowledge about pain in patients with haemophilia (PwH) is very restricted and mostly assessed by visual analogue scales or different pain questionnaires. However, both methods are unable to shed light on pain mechanisms in PwH and one might suspect that these methods are inappropriate to assess the pain condition comprehensively.
In a first study we could show that pain sensitivity thresholds are altered in PwH (Hilberg et al. 2011). By doing so, we were able to determine a reduced pressure pain threshold at knee and elbow joints of PwH in comparison to control subjects. This was not observed across reference points such as forehead and sternum. In a subsequent study (Krüger et al. 2018), which was supported by the clinical research grant program of the World Federation of Haemophilia (WFH), we detected a haemophilic specific pain profile in PwH. As a result, different alterations of the somatosensory system across both affected (the knees) and unaffected landmarks (the hand) in PwH were assessed for the first time. In particular, altered thermal as well as mechanical detection and pressure pain thresholds were examined in PwH when compared to healthy controls.
These findings exposed a dysfunction of fibres that detect warmth (C fibres), cold (Aδ fibres) and mechanical sensations (Aβ fibres). Based on the cross-sectional findings of a modified somatosensory system, it is not possible to determine the range of prevailing alterations in the pain condition caused by specific changes of progressing haemophilic arthropathy. Likewise, we can not verify the impact of bleeding events or factor replacement regime on the pain condition in PwH. Therefore, a comprehensive pain assessment during a one-year course will be conducted.